Many babies in the United States are born with a Patent Ductus Arteriosus (PDA), a vessel connecting the pulmonary artery and the aorta that doesn’t spontaneously close shortly after birth as it should. While a PDA typically closes on its own in full-term babies, small, premature newborns are more likely to require treatment and, at times, surgical ligation. The smallest and most preterm newborns are at highest risk for having a PDA that requires therapy to close.
In addition, there are roughly 60,000 preterm babies born each year in the United States who are delivered extremely preterm and classified as extremely low-birth-weight (ELBW) — weighing less than 2 pounds, 3 ounces. Of all newborns, these babies are at the highest risk of having a “hemodynamically significant PDA,” one that is large and causes symptoms. The presence of a hemodynamically significant PDA has been shown in some studies to be associated with complications such as severe chronic lung disease, intraventricular hemorrhage and necrotizing enterocolitis.
Determining the safest approach to PDA management in extremely preterm newborns is challenging. Many centers rely on medications to induce closure of a PDA, which may be contraindicated (potentially harmful) or ineffective.
Traditionally, surgical ligation was the only alternative to medical therapy to close a PDA. While effective, it involves an invasive procedure with several potential risks and complications. Historically, neonatologists and cardiologists lacked an alternative to medications and surgical ligation to close a hemodynamically significant PDA in tiny, preterm newborns.
Almost three years ago, the U.S. Food and Drug Administration approved Abbott Laboratories’ Amplatzer PiccoloTM Occluder. The device and procedure have been successfully used to close PDAs in ELBW preemies in a growing number of U.S. medical centers.
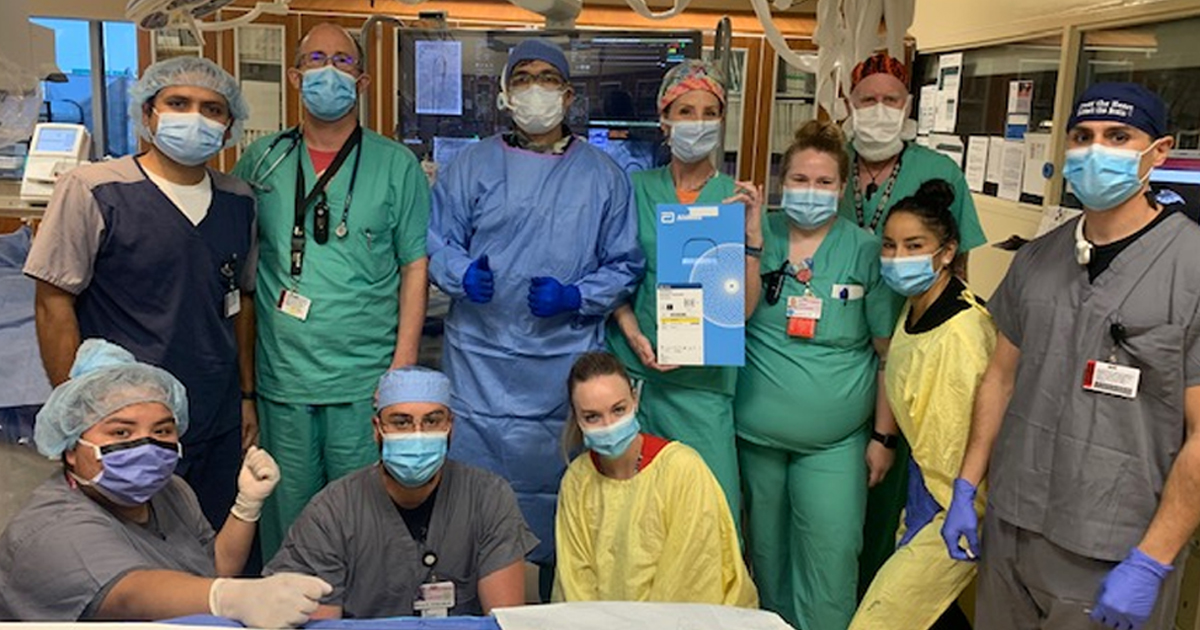
Adding to the list, this year, Presbyterian Hospital in Albuquerque, New Mexico, became the first in the state to perform PDA closures on micro-preemies (under 1 pound, 12 ounces at birth) using the Amplatzer Piccolo Occluder. At birth, the first baby weighed 540 grams (1 pound, 1 ounce) at 23 weeks and two days gestation. When the procedure was performed, he weighed 900 grams (1 pound, 9 ounces) and was 34 days old.
“Since its approval, the Piccolo device has become more widely used to avoid the need for surgical ligation; however, micro-preemies introduce a unique aspect to the procedure because of the size of the baby and the technique used for closing the PDA,” said Michael Rankin, M.D., neonatologist at Presbyterian Hospital in Albuquerque and neonatal transport director for Pediatrix® Medical Group of New Mexico. “This was the first Piccolo procedure performed on a baby that small and of that gestation in the state of New Mexico. The procedure, along with our close collaboration with Dr. Nasser Moiduddin and his team, is incredibly valuable to us, enabling us to relatively safely close a PDA on a symptomatic baby.”
A procedure with a tiny neonate requires tremendous collaboration among specialties from different areas within a hospital. Because this was the first of its kind at Presbyterian Hospital and precision was key to success, the assembled team performed several dry runs before the procedure to ensure each person knew exactly what to do and when to do it.
“The actual procedure isn’t terribly difficult or complicated,” said Nasser Moiduddin, M.D., the pediatric cardiologist at Presbyterian who performed New Mexico’s first Amplatzer Piccolo Occluder placement in a micro-preemie. “There’s an entire process around something like this to ensure everything goes smoothly in the cath lab. For example, we involve a mechanical engineer to adjust the temperature so it’s cool enough to avoid equipment failure but at the same time keeps the baby warm enough. The anesthesiologist, in addition to many other responsibilities, needs to ensure the baby stays warm throughout the procedure. It’s imperative that everyone in the cath lab can see the baby while he is draped, and we have a protocol in place to deal with anything that doesn’t go as planned. We even had security guards at the elevators to ensure the baby was transported back to the NICU as quickly and seamlessly as possible.”
There are numerous moving parts when it comes to transporting a micro-preemie to the cath lab, performing a heart procedure and transporting the baby back to the NICU. According to Dr. Moiduddin, the NICU team is critical before, during and after this type of procedure.
“The NICU team was instrumental in everything from A to Z,” he said. “There are a lot of things the NICU team takes care of before the baby ever reaches the cath lab, such as having the exact drugs and dosing readily available, intubating the baby, running the PICC line into the baby’s vein, providing baseline lab results, having an extra IV on hand, placing the blood pressure cuff and pulse oximeter and making sure the baby’s temperature is good. They also place the esophageal temperature probe, which helps me in terms of landmarks of where to place the device, and they help safely transport the baby to and from the cath lab. The bedside nurses, who know the baby better than anyone, are vital in alerting us of any potential issues that may stress the baby.”
Since the procedure on the first micro-preemie in New Mexico, Dr. Moiduddin has performed three more Piccolo procedures on micro-preemies, with the assistance of the NICU team and other hospital personnel. The first two cases included the same NICU team, including Dr. Rankin; Lisa Flowers, neonatal nurse practitioner; Stephanie Vargas, respiratory therapist; and Chelsea Cheshire, registered nurse.
“On behalf of the Presbyterian NICU team, we’re proud to have embarked on this journey with Dr. Moiduddin in securing successful outcomes for our tiniest, most fragile patients,” said Dr. Rankin. “And, together, we look forward to using these types of medical breakthroughs to give our patients every opportunity to thrive.”
Want to learn more about Prematurity Awareness Month or preterm babies?
Check out our other related articles:
Smaller than a Barbie doll, larger than life
Like mother, like daughter: A Christmas gift that spans decades
Leading the way for advancements in neonatal care
Caring for NICU babies: It takes a village
Five ways Graham’s Foundation provides support for premature infants
NNP cares for babies in the NICU, while the NICU cares for her preemies